Biogeochemical Cycles: Carbon, Water & Nitrogen
Atoms cycle through ecosystems endlessly. Master the carbon cycle (photosynthesis ↔ respiration), the water cycle (evaporation/precipitation), and the nitrogen cycle (fixation + denitrification).
Atoms recycle, energy doesn't
One of the deepest principles in ecology: energy flows in one direction (sun → producer → consumer → heat), but matter cycles. The same carbon atom in your body might have been in a dinosaur, a fern, or the atmosphere a billion years ago.
The CBE focuses on three cycles. Each has the same structure: reservoirs (where the element sits) and processes (how it moves between them).
The carbon cycle
Carbon is the backbone of every organic molecule. It moves between four main reservoirs:
- Atmosphere — as CO₂.
- Plants and animals — in glucose, proteins, fats, DNA.
- Soil and decomposers — in dead matter being broken down.
- Fossil fuels and ocean — long-term storage.
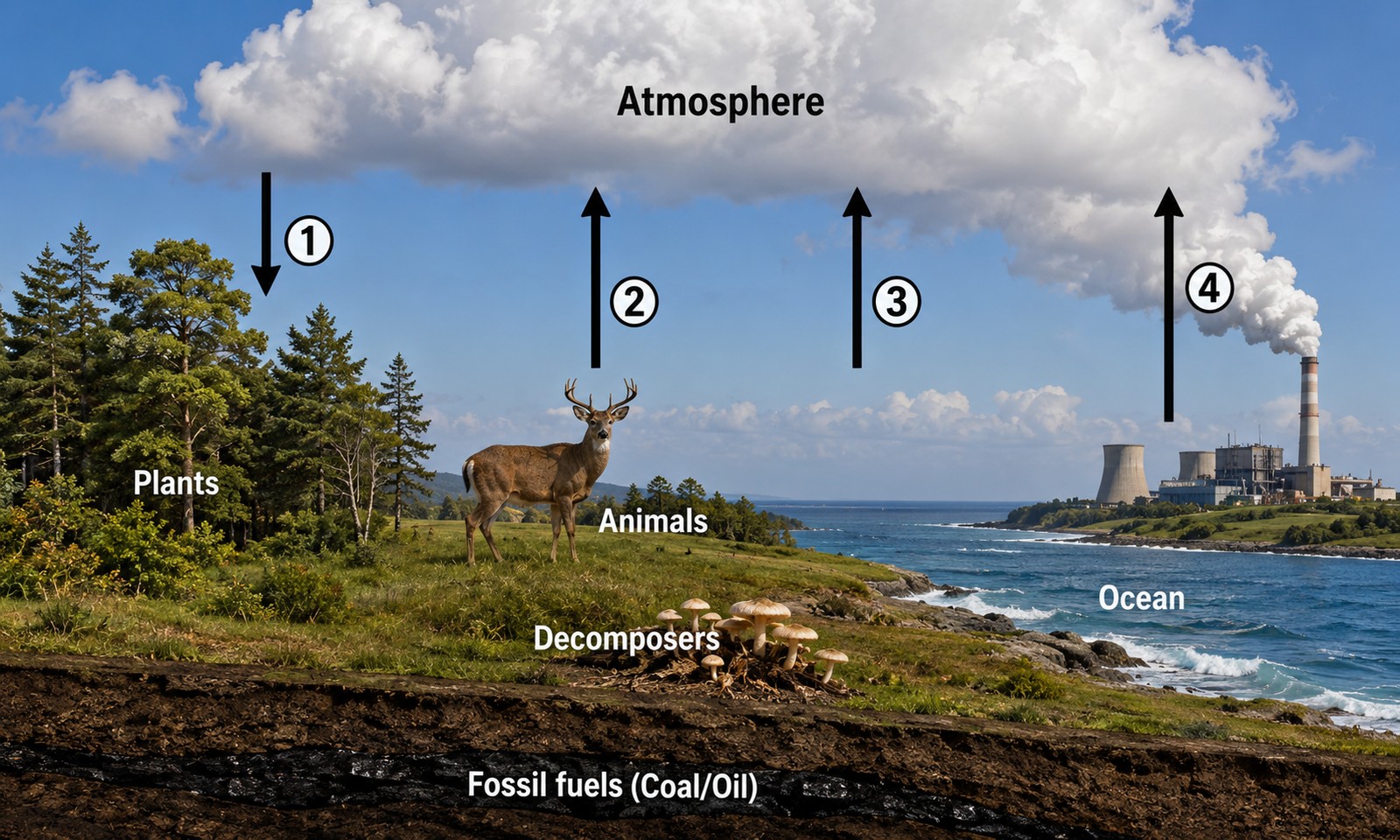
- Photosynthesis — pulls CO₂ from atmosphere into plant tissue. (Atmosphere → plants)
- Cellular respiration — releases CO₂ from animals/plants back to atmosphere. (Organisms → atmosphere)
- Decomposition — decomposers break down dead matter, releasing CO₂. (Decomposers → atmosphere)
- Combustion — burning fossil fuels or wood releases CO₂. (Fuel → atmosphere)
The water cycle
The water cycle has four major processes you should know:
- Evaporation — liquid water turns to vapor (mostly from oceans/lakes).
- Transpiration — water vapor released from plant leaves through stomata.
- Condensation — vapor cools and forms clouds.
- Precipitation — rain, snow, sleet falls back to surface.
The full circuit: ocean → evaporation → clouds → precipitation → rivers → ocean.
The nitrogen cycle
The atmosphere is 78% nitrogen gas (N₂), but plants and animals can’t use it directly. Bacteria do all the heavy lifting.
- Nitrogen fixation — bacteria convert atmospheric N₂ into usable ammonia (NH₃) or nitrate (NO₃−). Most nitrogen-fixing bacteria live in legume root nodules (peas, beans, clover).
- Nitrification — soil bacteria convert ammonia → nitrites → nitrates.
- Assimilation — plants absorb nitrates from soil and use them to build proteins and DNA.
- Decomposition (ammonification) — decomposers break down dead organic matter back into ammonia.
- Denitrification — soil bacteria convert nitrates back into atmospheric N₂. Closes the loop.
Bonus: mutualism in the nitrogen cycle
Legume plants (peas, beans, clover) host nitrogen-fixing bacteria in special root nodules. The bacteria get sugars from the plant; the plant gets usable nitrogen. Classic mutualism.